The atomic number of fluorine is 9 therefore, it possesses 9 electrons in its neutral atomic form. Start by calculating the number of valence electrons in each atom of F2 and see how many more electrons each fluorine atom needs to form an octet. Let us take a look at the chemical bonding represented by lewis structure in F2. This is exactly why they are called “noble.” 
Noble gases already have completely filled valance shells, so they do not need to bond/react with any other atoms/molecules. 
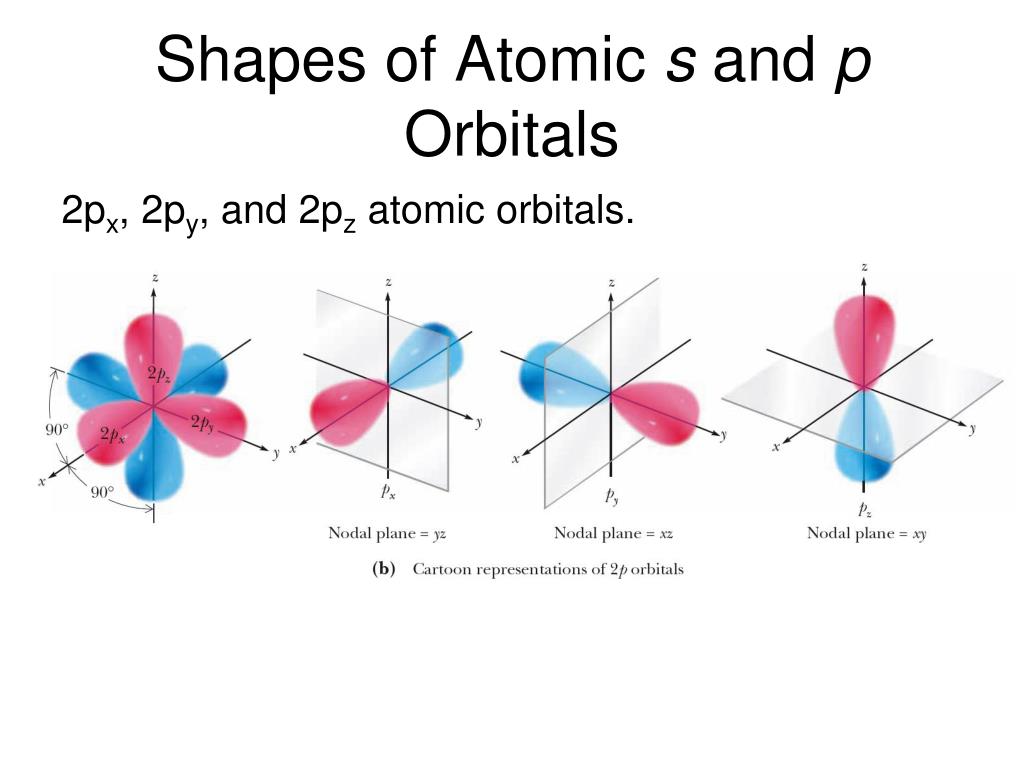
Hydrogen is an exception, though it seeks a duplet, not octet, because it has only one electron in its K shell, and thus needs only one more to achieve the maximum capacity of K shell. Several atoms tend to seek eight electrons in their valence shell through chemical bonding this is referred to as the octet rule and is reflected in the Lewis structure of a molecule. In a typical Lewis structure, each valence electron is represented as a dot, and a covalent bond between two atoms (formed as a result of sharing of two electrons) is represented as a line. It is to be noted though that this theory about the electronic structure is quite primitive and most limited. The valence electrons in each atom are the ones that participate in the bonding, and hence they are the only ones displayed in the Lewis structures. attenuators, filters, transducers and multiple state memory).The Lewis theory of chemical bonding helps us visualize the arrangement of atoms-how they are attached or bonded-in molecules. The types of materials we investigate have potential applications in energy conversion, transparent conducting oxides, gas sensors, catalysts, low temperature co-fired ceramics, dielectric ceramics, solid oxide fuel cells, and electronics (e.g. There is a continual need for: 1) a better understanding of composition-structure-property relationships, 2) new compounds/materials with improved properties, and 3) more economical compounds/materials. The results of the research are presented at conferences and in publications. Group members learn about conducting literature searches and manuscript writing. Measurements of their chemical and properties are conducted using electrical and dielectric characterization as a function of temperature and atmosphere, magnetic measurements, and photocatalytic properties. Characterization of their resulting phase assemblages and crystal structures is conducted through the use of X-ray diffraction techniques and crystallography. Approaches for materials design and discovery include a target approach using structure prediction, a directed approach using chemical intuition and substitutions, and exploratory through phase diagram development. In our research laboratory we utilize various solid state synthesis methods including conventional high temperature (up to 1600 ℃), flux, solvothermal, sealed tube and gas flow reactions. My research group is interested in the synthesis and characterization of solid-state inorganic materials and in fundamental structure/property relationships of solid state oxides. Solid-State, Inorganic, Materials Chemistry National Research Council Postdoctoral Fellow, (2003-2004) National Institute of Standards and Technology,NIST, Gaithersburg, MD of Chemistry and Biochemistry, University of South Carolina, Columbia, SC 
Inorganic Chemistry (1998-2002), Department of Chemistry, Ohio State UniversityĪssociate Professor (2010-present), Department of Chemistry, University of North Florida, Jacksonville, FLĪssistant Professor (2006-2010), Department of Chemistry, University of North Florida, Jacksonville, FL I then held a postdoctoral position with Professor Hanno zur Loye at the University of South Carolina, before starting at the University of North Florida.īS Chemistry (1994-1998), Department of Chemistry, Youngstown State University Following graduate school I spent two years as a National Research Council Postdoctoral Research Fellow at the National Institute of Standards and Technology in Gaithersburg MD, where I worked with Dr. My research advisor was Professor Patrick Woodward and my research at that time focused experimental solid state chemistry and on the development of a software program ( SPuDS) to calculate the crystal structures of perovskites. I obtained a BS in Chemistry from Youngstown State University in 1998.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
